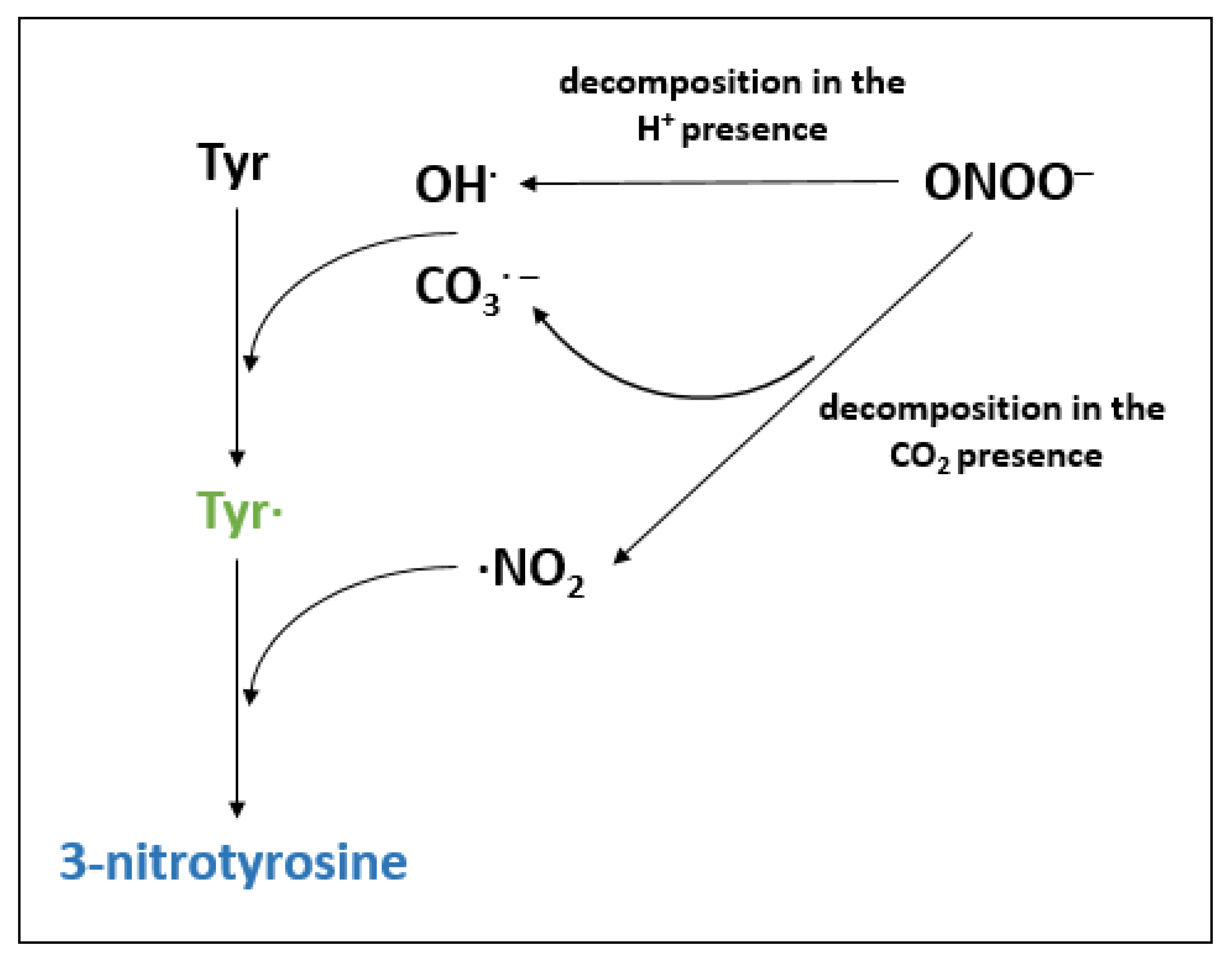
The species H2O2, ONOO⁻, NO, NO2⁻ being directly oxidized at their
€ 22.99 · 5 (585) · Auf Lager

Download scientific diagram | The species H2O2, ONOO⁻, NO, NO2⁻ being directly oxidized at their distinct potentials and their reconstructed fluxes. Reproduced from [2] with permission from Wiley from publication: Nanomaterial-based electrochemical sensors and optical probes for detection and imaging of peroxynitrite: a review | Peroxynitrite (PON for short) is a powerful nitrating, nitrosating and oxidative agent for cellular constituents. In vivo, PON is formed through the diffusion-controlled reaction between superoxide radical (O2•-) and nitric oxide (•NO). This critical review (with 67 refs.) | Electrochemical Sensors, Theranostics and Biocompatibility | ResearchGate, the professional network for scientists.

Hydrogen Peroxide (H2O2)- and Nitric Oxide (NO)-Derived

Hydrogen Peroxide (H2O2)- and Nitric Oxide (NO)-Derived
Solved 1. Microbial nitrification (the oxidation of ammonium

Oxidative stress and antioxidants
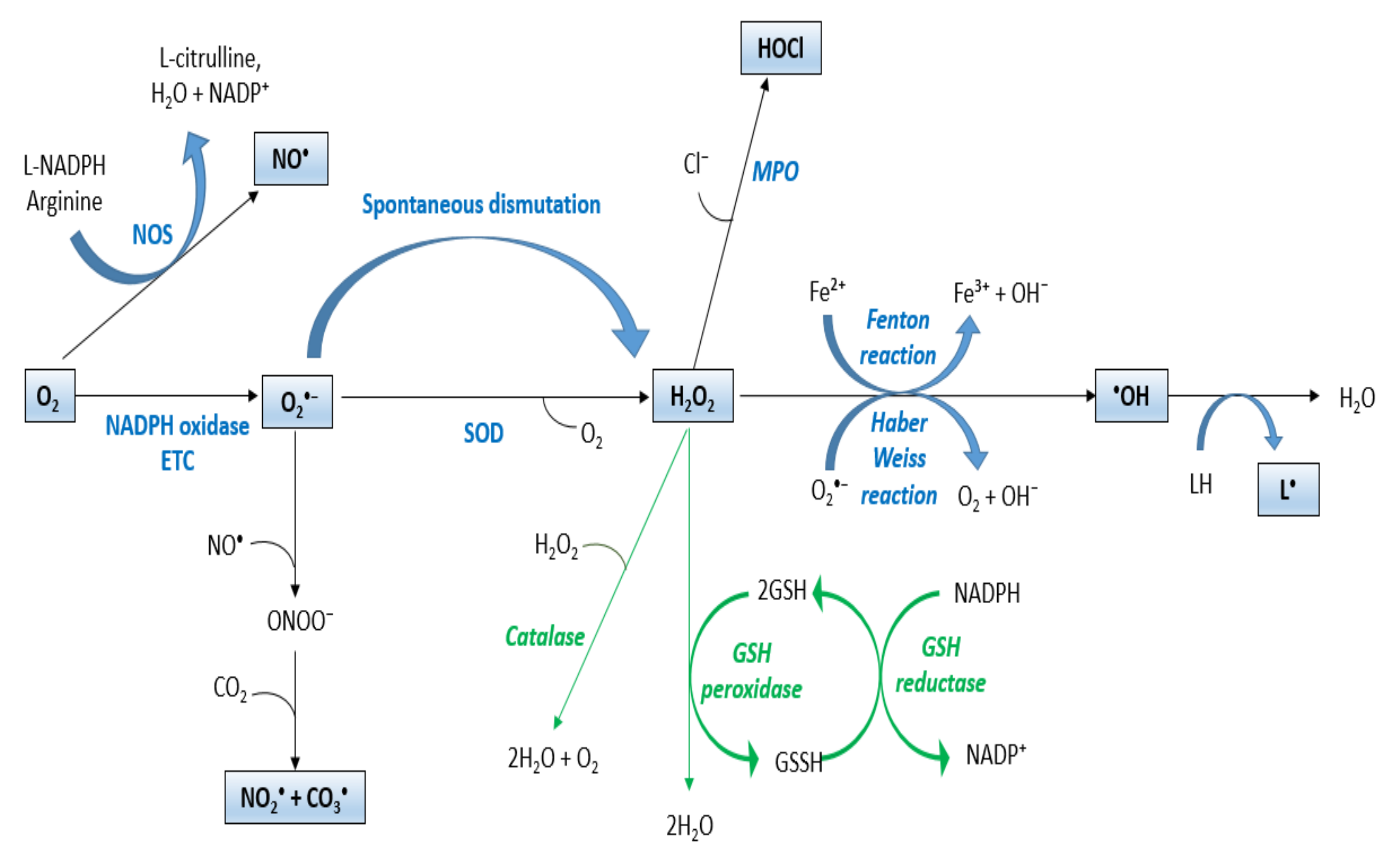
Antioxidants, Free Full-Text

PDF) The reaction of H2O2 with NO2 and NO
NO2 + H2O2 = HNO3 - Balanced Chemical Equation

Solved Sn032-t. Zn+ H2O+ 20H SnO22-+ Zn(OH)42. In the above

Solved Identify the species oxidized, the species reduced

Fenton-Mediated Oxidation in the Presence and Absence of Oxygen

PDF) The reaction of H2O2 with NO2 and NO

The species H2O2, ONOO⁻, NO, NO2⁻ being directly oxidized at their

Non-radical pathway dominated catalytic oxidation of As(III) with

Antioxidants, Free Full-Text
Hydrogen Peroxide (H2O2)- and Nitric Oxide (NO)-Derived